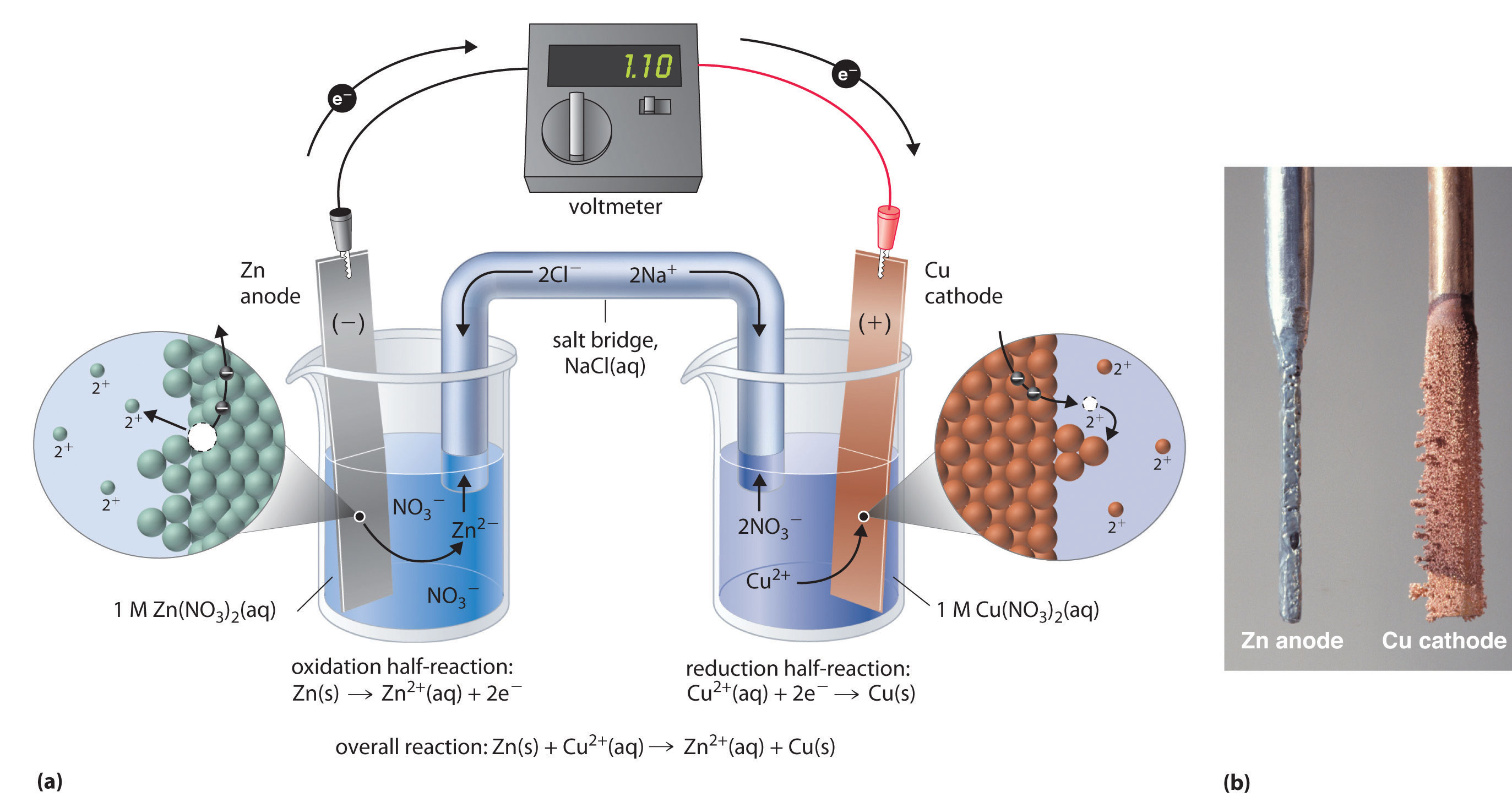
An animation from you tube explaining how the reaction occurs in a Daniell Cell
2013-07-15
2013-07-07
Electrolysis of Molten Lead(II) Bromide
 |
| Set-up of apparatus to study electrolysis of molten lead(II) bromide |
| Diagram shows reactions occurs during electrolysis of molten lead(II) bromide at both electrodes |
- Molten lead(II) bromide consists of lead(II) ions and bromide ions.
- During electrolysis of molten lead(II) bromide, bromide ions move to anode and lead(II) ions move to cathode.
- At anode, bromide ions are discharged by releasing electrons to form bromine. Half equation for the reaction is 2Br- --> Br2 + 2e-
- At cathode, lead(II) ions are discharged by accepting electrons to form lead. Half equation for the reaction is
Subscribe to:
Posts (Atom)